On the road to Munich in January for DLD14, the 10th annual DLD conference (Digital-Life-Day) run by Steffi Czerny and Lukas Kubina for Hubert Burda Media. The theme this year: "Content and Context." It was the fifth time Edge has been asked to participate. (See below for links to our previous DLD co-events.)

Steffi Czerny and John Brockman
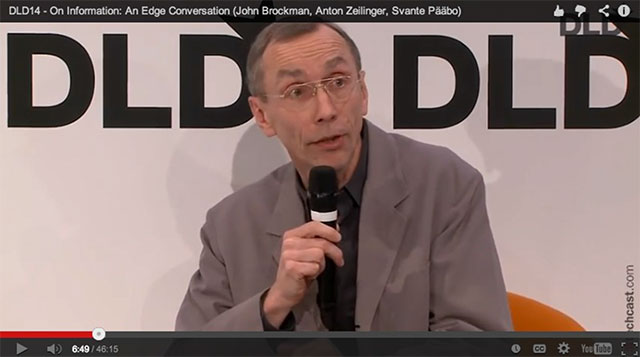
This year the Edge conversation was on "information" from the Neandertal DNA sequenced by Svante Pääbo, the founder of the field of ancient DNA, to the multi-particle entanglement states of physicist Anton Zeilinger, which have become essential in fundamental tests of quantum mechanics and in quantum information science, most notably in quantum computation. In addition, Edge's roving editor, Jennifer Jacquet, was present for a session on "Time's Role in the Tragedy of the Commons" in which she developed themes in her work recently presented on Edge.
Svante Pääbo, Anton Zeilinger, John Brockman: On Information
Information is the foundation of our universe—and life itself. Cultural impresario John Brockman hosts a Third Culture conversation, spanning science and the humanities.
SVANTE PÄÄBO the founder of the field of ancient DNA, is Director, Department of Genetics, Max Planck Institute for Evolutionary Anthropology, Leipzig. Among his achievements are the first demonstration of DNA survival in an ancient Egyptian mummy, the first amplification of ancient DNA, the first study of the DNA from the Iceman found in the Alps, and the first retrieval of DNA from a Neanderthal in 1997. Four years ago, he initiated and organized an effort to sequence the entire Neanderthal genome. The first scientific overview of the genome was published in 2009 and was front page news wordwide. He is the author of Neanderthal Man: In Search of Lost Genomes. Svante Pääbo's Edge Bio Page
ANTON ZEILINGER, a physicist, is Professor of Physics at the Quantum Optics, Quantum Nanophysics, Quantum Information Institute of University of Vienna. He is President of the Austrian Academy of Sciences and the author of Dance of the Photons: From Einstein to Quantum Teleportation. Zeilinger is a pioneer in the field of quantum information and of the foundations of quantum mechanics. He realized many important quantum information protocols for the first time, including quantum teleportation of an independent qubit, entanglement swapping (i.e. the teleportation of an entangled state), hyper-dense coding (which was the first entanglement-based protocol ever realized in experiment), entanglement-based quantum cryptography, one-way quantum computation and blind quantum computation. His further contributions to the experimental and conceptual foundations of quantum mechanics include multi-particle entanglement and matter wave interference all the way from neutrons via atoms to macromolecules such as fullerenes. Anton Zeilinger's Edge Bio Page
Jennifer Jacquet: Times Role in the Tragedy of the Commons
How do tensions between individuals and groups play out? Between high-consuming people and low? Between the now and the future? Game theory offers answers.
JENNIFER JACQUET is Clinical Assistant Professor of Environmental Studies, NYU, researching shame, cooperation and the tragedy of the commons. She is also Edge's Roving Editor (see interviews with Adam Alter and Joseph Heinrich).
Her work was recently featured on Edge after Nature Climate Change published a study by Jacquet and her colleagues at two Max Planck Institutes on "Delayed Gratification Hurts Cimate Change Cooperation". Jennifer Jacquet's Edge Bio Page
EDGE @DLD: 2011: AN EDGE CONVERSATION IN MUNICH with Stewart Brand, George Dyson, Kevin Kelly, introduced by Andrian Kreye. A session with three of the original members of Edge who year in and year out provide the core sounding board for the ideas and information we present to the public. I refer to them in private correspondance as "The Council." Every year, beginning late summer, I consult with Stewart Brand, Kevin Kelly, and George Dyson, and together we create the Edge Annual Question which Edge has been asking since 1996. 2010: "INFORMAVORE" with Frank Schirrmacher, Editor of the Feuilleton and Co-Publisher of Frankfurter Allgemeine Zeitung, Andrian Kreye, Feuilleton Editor of Sueddeutsche Zeitung, Munich; and Yale computer science visionary David Gelernter, who, in his 1991 book Mirror Worlds presented what's now called "cloud computing." 2009: REFLECTIONS ON A CRISIS: Daniel Kahneman, the greatest living psychologist, and Nassim Nicholas Taleb, the foremost scholar of extreme events discuss hindsight biases, the illusion of patterns, perception of risk, and denial. 2008: LIFE: A GENE-CENTRIC VIEW: Richard Dawkins & J. Craig Venter. It's not everyday you have Richard Dawkins and Craig Venter on a stage talking for an hour. That it occured in Germany, where the culture had been resistant to open discussion of genetics, and at DLD, the Digital, Life, Design conference organized by Hubert Burda Media in Munich, a high-level event for the digital elite—the movers and shakers of the Internet—made the discussion particularly interesting.
In July, 2013, Edge invited a group of social scientists to participate in an Edge Seminar at Eastover Farm focusing on the state of the art of what the social sciences have to tell us about human nature. The ten speakers were Sendhil Mullainathan, June Gruber, Fiery Cushman, Rob Kurzban, Nicholas Christakis, Joshua Greene, Laurie Santos, Joshua Knobe, David Pizarro, Daniel C. Dennett. Also participating were Daniel Kahneman, Anne Treisman, Jennifer Jacquet.
We asked the participants to consider the following questions:
"What's new in your field of social science in the last year or two, and why should we care?" "Why do we want or need to know about it?" "How does it change our view of human nature?"
And in so doing we also asked them to focus broadly and address the major developments in their field (including but not limited to their own research agenda). The goal: to get new, fresh, and original up-to-date field reports on different areas of social science.
What Big Data Means For Social Science (Sendhil Mullainathan) | The Scientific Study of Positive Emotion (June Gruber) | The Paradox of Automatic Planning (Fiery Cushman) | P-Hacking and the Replication Crisis (Rob Kurzban) | The Science of Social Connections (Nicholas Christakis) | The Role of Brain Imaging in Social Science (Joshua Greene) | What Makes Humans Unique (Laurie Santos) | Experimental Philosophy and the Notion of the Self (Joshua Knobe) | The Failure of Social and Moral Intuitions (David Pizarro) | The De-Darwinizing of Cultural Change (Daniel C. Dennett)
HeadCon '13: WHAT'S NEW IN SOCIAL SCIENCE was also an experiment in online video designed to capture the dynamic of an Edge seminar, focusing on the interaction of ideas, and of people. The documentary film-maker Jason Wishnow, the pioneer of "TED Talks" during his tenure as director of film and video at TED (2006-2012), helped us develop this new iteration of Edge Video, filming the ten sessions in split-screen with five cameras, presenting each speaker and the surrounding participants from multiple simultaneous camera perspectives.
We are now pleased to present the program in its entirety, nearly six hours of Edge Video and a downloadable PDF of the 58,000-word transcript.
The great biologist Ernst Mayr (the "Darwin of the 20th Century") once said to me: "Edge is a conversation." And like any conversation, it is evolving. And what a conversation it is!
(6 hours of video; 58,000 words)
John Brockman, Editor
Russell Weinberger, Associate Publisher

Download PDF of Manuscript | Continue to Video and Online Text
Sendhil Mullainathan: What Big Data Means For Social Science (Part I)
We've known big data has had big impacts in business, and in lots of prediction tasks. I want to understand, what does big data mean for what we do for science? Specifically, I want to think about the following context: You have a scientist who has a hypothesis that they would like to test, and I want to think about how the testing of that hypothesis might change as data gets bigger and bigger. So that's going to be the rule of the game. Scientists start with a hypothesis and they want to test it; what's going to happen?
June Gruber: The Scientific Study of Positive Emotion (Part II)
What I'm really interested in is the science of human emotion. In particular, what's captivated my field and my interest the most is trying to understand positive emotions. Not only the ways in which perhaps we think they're beneficial for us or confer some sort of adaptive value, but actually the ways in which they may signal dysfunction and may not actually, in all circumstances and in all intensities, be good for us.
June Gruber is Assistant Professor of Psychology, Director, Positive Emotion & Psychopatology Lab, Yale University.
Fiery Cushman: The Paradox of Automatic Planning (Part III)
I want to tell you about a problem that I have because it highlights a deep problem for the field of psychology. The problem is that every time I sit down to try to write a manuscript I end up eating Ben and Jerry's instead. I sit down and then a voice comes into my head and it says, "How about Ben and Jerry's? You deserve it. You've been working hard for almost ten minutes now." Before I know it, I'm on the way out the door.
Fiery Cushman is Assistant Professor, Cognitive, Linguistic, Social Science, Brown University.
Rob Kurzban: P-Hacking and the Replication Crisis (Part IV)
The first three talks this morning I think have been optimistic. We've heard about the promise of big data, we've heard about advances in emotions, and we've just heard from Fiery, who very cleverly managed to find a way to leave before I gave my remarks about how we're understanding something deep about human nature. I think there's a risk that my remarks are going to be understood as pessimistic but they're really not. My optimism is embodied in the notion that what we're doing here is important and we can do it better.
Rob Kurzban is an Associate Professor, University of Pennsylvania specializing in evolutionary psychology: Author, Why Everyone (Else) Is A Hypocrite.
Nicholas Christakis: The Science of Social Connections (Part V)
If you think about it, humans are extremely unusual as a species in that we form long-term, non-reproductive unions to other members of our species, namely, we have friends. Why do we do this? Why do we have friends? It's not hard to construct an argument as to why we would have sex with other people but it's rather more difficult to construct an argument as to why we would befriend other people. Yet we and very few other species do this thing. So I'd like to problematize that, I'd like to problematize friendship first.
Nicholas Christakis is a Physician and Social Scientist; Director, The Human Nature Lab, Yale University; Coauthor, Connected: The Surprising Power Of Our Social Networks And How They Shape Our Lives.
Joshua Greene: The Role of Brain Imaging in Social Science (Part VI)
We're here in early September 2013 and the topic that's on everybody's minds, (not just here but everywhere) is Syria. Will the U.S. bomb Syria? Should the U.S. bomb Syria? Why do some people think that the U.S. should? Why do other people think that the U.S. shouldn't? These are the kinds of questions that occupy us every day. This is a big national and global issue, sometimes it's personal issues, and these are the kinds of questions that social science tries to answer.
Joshua Greene is John and Ruth Hazel Associate Professor of the Social Sciences and the director of the Moral Cognition Laboratory in the Department of Psychology, Harvard University. Author, Moral Tribes: Emotion, Reason, And The Gap Between Us And Them.
Laurie Santos: What Makes Humans Unique (Part VII)
The findings in comparative cognition I'm going to talk about are often different than the ones you hear comparative cognitive researchers typically talking about. Usually when somebody up here is talking about how animals are redefining human nature, it's cases where we're seeing animals being really similar to humans—elephants who do mirror self-recognition; rodents who have empathy; capuchin monkeys who obey prospect theory—all these cases where we see animals doing something really similar.
Laurie Santos is Associate Professor, Department of Psychology; Director, Comparative Cognition Laboratory, Yale University.
Joshua Knobe: Experimental Philosophy and the Notion of the Self (Part VIII)

What is the field of experimental philosophy? Experimental philosophy is a relatively new field—one that just cropped up around the past ten years or so, and it's an interdisciplinary field, uniting ideas from philosophy and psychology. In particular, what experimental philosophers tend to do is to go after questions that are traditionally associated with philosophy but to go after them using the methods that have been traditionally associated with psychology.
Joshua Knobe is an Experimental Philosopher; Associate Professor of Philosophy and Cognitive Science, Yale University.
We had people interact—strangers interact in the lab—and we filmed them, and we got the cues that seemed to indicate that somebody's going to be either more cooperative or less cooperative. But the fun part of this study was that for the second part we got those cues and we programmed a robot—Nexi the robot, from the lab of Cynthia Breazeal at MIT—to emulate, in one condition, those non-verbal gestures. So what I'm talking about today is not about the results of that study, but rather what was interesting about looking at people interacting with the robot.
David Pizarro is Associate Professor of Psychology, Cornell University, specializing in moral judgement.
Daniel C. Dennett: The De-Darwinizing of Cultural Change (Part X)
Think for a moment about a termite colony or an ant colony—amazingly competent in many ways, we can do all sorts of things, treat the whole entity as a sort of cognitive agent and it accomplishes all sorts of quite impressive behavior. But if I ask you, "What is it like to be a termite colony?" most people would say, "It's not like anything." Well, now let's look at a brain, let's look at a human brain—100 billion neurons, roughly speaking, and each one of them is dumber than a termite and they're all sort of semi-independent. If you stop and think about it, they're all direct descendants of free-swimming unicellular organisms that fended for themselves for a billion years on their own. There's a lot of competence, a lot of can-do in their background, in their ancestry. Now they're trapped in the skull and they may well have agendas of their own; they have competences of their own, no two are alike. Now the question is, how is a brain inside a head any more integrated, any more capable of there being something that it's like to be that than a termite colony? What can we do with our brains that the termite colony couldn't do or maybe that many animals couldn't do?
Daniel C. Dennett is a Philosopher; Austin B. Fletcher Professor of Philosophy, Co-Director, Center for Cognitive Studies, Tufts University; Author, Intuition Pumps.
ALSO PARTICIPATING

Daniel Kahneman is Recipient, Nobel Prize in Economics, 2002; Presidential Medal of Freedom, 2013; Eugene Higgins Professor of Psychology, Princeton University; Author, Thinking Fast And Slow. Anne Treisman is Professor Emeritus of Psychology, Princeton University; Recipient, National Medal of Science, 2013.

Jennifer Jacquet is Clinical Assistant Professor of Environmental Studies, NYU; Researching cooperation and the tragedy of the commons.
Out-take from the trailer I made for the 1968 movie "Head" (Columbia Pictures; Directed by Bob Rafelson; Written by Jack Nicholson)

DANIEL KAHNEMAN & RICHARD THALER
at Edge Retreat, August 22, 2013

[expand]








Edge Master Class 2011: "What's New In Human Nature"
You can answer the question, but are you bright enough to ask it?

INTRODUCTION
by John Brockman
The Edge motto, adopted from the artist James Lee Byars' "World Question Center" is: "To arrive at the edge of the world's knowledge, seek out the most complex and sophisticated minds, put them in a room together, and have them ask each other the questions they are asking themselves." As Wallace Stevens wrote in "Notes Toward A Supreme Fiction" (1942): "The final elegance, not to console / Nor sanctify, but plainly to propound."
It's the quality of the questions that defines the scientific endeavor, not the answers encrusted in stories and narratives. This year, at SciFoo 2013, Edge presented the opportunity to several of the nearly 300 participants to respond to the following: "What is your question from SciFoo 2013?" Watch the 8-minute video below for the responses. This is followed by responses to the question "Who and/or what was fresh and new at SciFoo 2013?", and a photo gallery, which extends to San Francisco the following evening.
But first, what is "SciFoo"?
The annual event is run by three sponsors: O'Reilly Media (Tim O'Reilly is responsible for "FOO," or, "friends of O'Reilly"), Nature magazine (and their spin-off company, Digital Science), and Google. This year 62% of the participants were new. This approach keeps the event new and fresh every year. The ratio of participants to interesting people? 1-to-1.
As theoretical physicist and Nobel Laureate Frank Wilczek noted in his report on SciFoo 2007 for Edge:
"SciFoo is a conference like no other. It brings together a mad mix from the worlds of science, technology, and other branches of the ineffable Third Culture at the Google campus in Mountain View. Improvised, loose, massively parallel—it's a happening. If you're not overwhelmed by the rush of ideas then you're not paying attention."
Also, see George Dyson's report on SciFoo 2007; Photos and comments on SciFoo 2009, and the Edge-SciFoo report on SciFoo 2011.
Tyler Cowen, Joseph "Yossi" Vardi, Carl Page, Fiery Cushman, Lee Smolin, Linda Stone, Paul Davies, Paul Steinhardt, Peter Norvig, Richard Potts, Steve Fuller, Stuart Firestein
WHO AND/OR WHAT WAS FRESH AND NEW AT SCI/FOO 2013?
George Dyson, Esther Dyson, Steve Fuller, Stewart Brand, Paul Saffo, Coco Krumme, John Coates
A fascinating weekend, with one lingering regret—that I missed Rory Wilson’s talk on the use of accelerometers in free living animals. I had to piece together the gist of his talk from scraps given me by others and, fortunately, by Rory himself. In his work as an animal behaviorist Rory has been using actigraphy and accelerometers attached to animals in the wild in order to monitor continuously their energy expenditure. Beyond this, though, the data Rory has collected has uncovered subtle changes in background movement that actually reflect the animal’s motivational state. This finding is tremendously interesting. As we learn more about the brain, the more we see that it is built primarily to plan and execute movement, that activities we—under the influence of our Platonic heritage—commonly viewed as pure thought in fact have a somatic echo. Some scientists working with actigraphy have even found predictors, tremors if you will, of impending neuro-degenerative disease. But background activity changes that may reflect motivational states? The very possibility should tantalize any behavioural scientist.
Serendipity was at work this weekend: I met perhaps the only other person at scifoo (on the entire googolplex?) with a dumb phone. I stubbornly hold onto mine for the very purpose of serendipity, to dampen distraction. Ziyad Marar and I had a wonderful conversation about habits and human behavior, and soon discovered we have in common 1999-era telephones, as well as a healthy skepticism of social media. I hadn't heard of Ziyad's books (Happiness Paradox, Deception, Intimacy), but I picked up two at scifoo and couldn't put them down: his writing mulls human nature, weaving in literature and philosophy without succumbing to the tired conventions of contemporary science writing. A breath of fresh air.
PAUL SAFFO: Kröpelin's Mysteries of the Sahara
The single most astonishing session for me was Stefan Kröpelin's "Miracle of the Sahara." Kroplin compressed 40 years of research and exploration into a whirlwind tour of the Sahara's mysteries and what it is like to do science there. Summarizing conditions, he noted, "Sometimes, you get stuck hundreds of times per day… and the real problem isn't the heat; it's the cold." The size of the U.S, Kröpelin's Sahara is full of mysteries: Gilf Kebir, a sand plateau atop an ancient fluvial system in Southwest Egypt holds rock art from the middle Holocene, over 10,000 figures in one cave alone. The Wadi Howar was thought by Heroditus to be the source of the Nile; now it is vast desert, but in it's heart is the Ounianga Kebir, a cluster of freshwater aquifer-fed "gravity lakes," and home to seven crocodiles, the remnant of an Ice Age population, now isolated from other crocs by hundreds of miles of searing desert.
But the biggest mystery was sitting right in front of Kröpelin as he spoke. It was a chunk of "Libyan Desert Glass," 28 million year-old fused glass the color of pale emerald and the purest natural glass in the world. Discovered by Europeans in 1932, the stuff is strewn across a vast area of desert at the edge of Egypt's great Sand Sea. Kröpelin's chunk looked like a glass meteorite, complete with ablation regmaglypts, suggesting that the glass was created by a meteor strike that liquefied the surface rocks in a process not unlike that which created tektite strewn-fields elsewhere on the earth. Except… no one has found a crater. Perhaps the glass is a radiative melt artifact of a Tunguska-like airburst? Others speculate that it is hydrovolcanic in origin, but no has found a volcanic source.
What we do know is that the Sahara's earliest inhabitants knapped the glass into tools, and that it was prized by the Egyptians—a scarab carved from the stuff is set into a pectoral worn by Tutankhamun. That fact tied nicely with Kröpelin's final and most surprising statement: the origins of the pharonic tradition lie in the Western Desert, not the Nile Valley. And if the Egyptian civilization was born from the Sahara, then by association, Europe's roots are hidden there as well.
STEWART BRAND: What if climate change is good for civilization?
(If it is, it would completely invalidate the first chapter of my Whole Earth Discipline, which spells out how climate change is an apocalypse-in-waiting for civilization. My personal mantra tries to be: "Every day I wonder how many things I am dead wrong about." This might be one of them.)
Climate change apparently ignited civilization in Egypt, said Stefan Kröpelin in a small, intense session at SciFoo. His four decades of insanely adventurous researches in the eastern Sahara—where no one goes—show that people lived all over that region when it was a rich savanna filled with game, before 8,000 years ago. In those centuries people avoided the Nile, which was dangerous with floods and fearsome animals.
In the period 5300-3500 BCE the Sahara gradually but relentlessly dried up. The people from the desert were forced to take on the Nile, first for farming, soon for a river-taming civilization of such durability that it eventually inspired Greece, Rome, and Europe.
Climate change made it happen. The evidence is in the sumptuous cave art at Wadi Sura and the silt of the astonishingly ancient Ounianga Lakes.
What is our unthinkably dangerous Nile that climate change might force us to take on? What abilities might we be forced to acquire?
STEVE FULLER: How must we re-orient ourselves to make the most out of seasteading?
At Sci Foo Camp 2013, David Ewing Duncan and Linda Avey presented a very persuasive case for an indefinite expansion of 'seasteading', a term that I had learned about from Duncan in an earlier session on cognitive and moral enhancement by neuroscientific means. I had been already familiar with Peter Thiel's support for a ship floating just beyond US territorial waters near San Francisco Bay that enabled innovators lacking US citizenship to conduct research outside the gaze of American regulators. However, the session enabled me to see that seasteading, far from being a strategy for avoiding inconvenient forms of regulation, on the contrary might be something that states themselves encourage—though perhaps indirectly, depending on the political climate. However, this proposal would make policy sense only on the following condition: The outcomes of the research conducted in these 'ethics-free' zones would have to be made public, with the understanding that no one would be prosecuted, no matter how bad the outcomes are perceived to have been. In other words, we would need to become sufficiently mature to accept the admission of error in pursuit of a good cause as adequate punishment in itself.
The most wonderful session I attended—and the most meaningful experience overall—was Michael Chorost talking about his own cochlear implant. It was what science really is—the response to curiosity. He told us how it worked, played recordings so we could get some sense of how things sound to people who have an implant. He passed some samples around the room for us to touch and examine. We talked about learning to hear—and how there's a point in childhood after which it gets harder and harder to learn. We got an understanding of the technology, and also of how the technology changes both individual lives and cultural norms—such as sign language, which may become the language of the poor deaf as the rich deaf start using cochlear implants. In theory they can be covered by Medicare, but somehow the rich seem to get them and the poor don't... Is that right?
Is there something similar coming for vision? (Of course, this comment—and the session itsel —raises questions and doesn't answer them all; that's the point of SciFoo...)
This was SciFoo's seventh year, and, in era where the half-life of a conference is measured in years, not decades, it is holding up well. Tim O'Reilly (O'Reilly), Timo Hannay (Nature/Digital Science )and Chris DiBona (Google) have found a formula that works, and are sticking to it. Big Data, once the all-consuming subject, is now just another scientific instrument, like a space telescope or an electron microscope, and the excitement was back to the details of what you can do with it, now that you have it. And how do you encourage scientific thinking in children (and politicians)? Among the people who showed up from left field and stole the show (a regular occurrence at SciFoo) this year was Carmen Medina, who, in her own introductory words, "spent 32 years at the Central Intelligence Agency and decided early on that how we form opinions is much more interesting than any particular opinion. Passionate about empowering heretics in the workplace and attacking conventional wisdom. Believe we need entirely new construct for the concept of national security, perhaps abandoning it altogether. Convinced there is worldwide conspiracy for preservation of Mediocrity." Amen!
Recently we have published a number of Conversations on related subjects such as "Big Data", "Linked Data", "Data Science", "Web Science", "Semantic Web", "Network Science". Clearly, a new realm is rapidly coming into public consciousness.
In this regard, we have set up this "Special Event" page on "Computational Social Science" to organize and present this material to our readers and to provide access to the ongoing Edge Conversations and related discussions.
Published to date are eight Conversations with: Dirk Helbing, Nicholas A. Christakis, J. Craig Venter, J. Craig Venter, Cesar Hidalgo, Sandy Pentland, Albert-László Barabási and Tim O'Reilly. The presentations include more than five hours of video as well as the texts.
—John Brockman
Editor
"THE CLOTHESLINE PARADOX"
A Conversation with Tim O'Reilly [10.4.12]

If we're going to get science policy right, it's really important for us to study the economic benefit of open access and not accept the arguments of incumbents. Existing media companies claim that they need ever stronger and longer copyright protection and new, draconian laws to protect them, and meanwhile, new free ecosystems, like the Web, have actually led to enormous wealth creation and enormous new opportunities for social value. And yes, they did in fact lead in some cases to the destruction of incumbents, but that's the kind of creative destruction that we should celebrate in the economy. We have to accept that, particularly in the area of science, there's an incredible opportunity for open access to enable new business models.
TIM O'REILLY is the founder and CEO of O'Reilly Media, Inc., a leading computer book publisher. O'Reilly Media also hosts conferences on technology topics, including the O'Reilly Open Source Convention, the Strata series of conferences on big data, and Tools of Change for Publishing. O'Reilly Media's Maker Media unit publishes Make Magazine and operates Maker Faire, the world's largest gathering of DIY hardware enthusiasts and entrepreneurs. O'Reilly AlphaTech Ventures is a leading early stage venture capital firm.
THINKING IN NETWORK TERMS
A Conversation with Albert-lászló Barabási [9.24.12]

One question that fascinated me in the last two years is, can we ever use data to control systems? Could we go as far as, not only describe and quantify and mathematically formulate and perhaps predict the behavior of a system, but could you use this knowledge to be able to control a complex system, to control a social system, to control an economic system?
ALBERT-LÁSZLÓ BARABÁSI is a Distinguished University Professor at Northeastern University, where he directs the Center for Complex Network Research, and holds appointments in the Departments of Physics, Computer Science and Biology, as well as in the Department of Medicine, Harvard Medical School and Brigham and Women Hospital, and is a member of the Center for Cancer Systems Biology at Dana Farber Cancer Institute.
Albert-László Barabási Edge Bio Page
REINVENTING SOCIETY IN THE WAKE OF BIG DATA
A Conversation with Alex (Sandy) Pentland [8.30.12]

With Big Data we can now begin to actually look at the details of social interaction and how those play out, and are no longer limited to averages like market indices or election results. This is an astounding change. The ability to see the details of the market, of political revolutions, and to be able to predict and control them is definitely a case of Promethean fire --- it could be used for good or for ill, and so Big data brings us to interesting times. We're going to end up reinventing what it means to have a human socie
ALEX 'SANDY' PENTLAND is a pioneer in big data, computational social science, mobile and health systems, and technology for developing countries. He is one of the most-cited computer scientists in the world and was named by Forbes as one of the world's seven most powerful data scientists. He currently directs the
Sandy Pentland's Edge Bio Page
WHAT IS VALUE? WHAT IS MONEY?
A Conversation with Cesar Hidalgo [8.28.12]
We have always had this tension of understanding the world, at small spatial scales or individual scales, and large macro scales. In the past when we looked at macro scales, at least when it comes to many social phenomena, we aggregated everything. Our idea of macro is, by an accident of history, a synonym of aggregate, a mass in which everything is added up and in which individuality is lost. What data at high spatial resolution, temporal resolution and typological resolution is allowing us to do, is to see the big picture without losing the individuality inside it.
CESAR HIDALGO is an assistant professor at the MIT Media Lab, and faculty associate at Harvard University’s Center for International Development. His work focuses on improving the understanding of systems by using and developing concepts of complexity, evolution, and network science. He is also the founder and driving force behind Cambridge Nights, a series of online video interviews with academics who discuss the way in which they view the world.
A NEW KIND OF SOCIAL SCIENCE FOR THE 21st CENTURY
A Conversation with Nicholas A. Christakis [8.21.12]
These three things—a biological hurricane, computational social science, and the rediscovery of experimentation—are going to change the social sciences in the 21st century. With that change will come, in my judgment, a variety of discoveries and opportunities that offer tremendous prospect for improving the human condition.
It's one thing to say that the way in which we study our object of inquiry, namely humans, is undergoing profound change, as I think it is. The social sciences are indeed changing. But the next question is: is the object of inquiry also undergoing profound change? It's not just how we study it that's changing, which it is. The question is: is the thing itself, our humanity, also changing?
NICHOLAS A. CHRISTAKIS is a Physician and Social Scientist, Harvard University; Coauthor (with James Fowler) of Connected: The Surprising Power of Our Social Networks and How They Shape Our Lives.
Nicholas A. Chrsitakis's Edge Bio Page
[40:59 minutes]
BIOLOGY AT THE SPEED OF LIGHT
After Dinner Talk by J. Craig Venter [7.10.12]
We can now send biology at the speed of light, and this is one of the implications of our work, which we recorded two years ago making the first synthetic life form. We completely synthesized the genetic code of a cell starting with a digital code in the computer—it's the ultimate interface between computers and biology. The digital code and the genetic code have a lot in common; something Schrodinger pointed out in 1943, saying it could be something as simple as the Morse code. ... Digital code, as you know, is a binary code, and ones and zeroes, and your genetic code is literally four-base code with ACGs and Ts. We can now readily convert in between the two, and we can define life at its most basic level. Things that were a mystery fifty, sixty, seventy years ago, we now understand completely.
Genomics researcher J. CRAIG VENTER is regarded as one of the leading scientists of the 21st century, most notably for the first sequencing and analysis of the human genome published in 2001 and the most recent and most complete sequencing of his diploid human genome in 2007. He is Co-Founder, Chairman, Synthetic Genomics, Inc.; Founder, J. Craig Venter Institute; Author, A Life Decoded.
J. Craig Venter's Edge Bio Page
[34:16 minutes]
WHAT IS LIFE? A 21st CENTURY PERSPECTIVE
On the 70th Anniversary of Schroedinger's Lecture at Trinity College by J. Craig Venter [7.12.12]
I view DNA as an analogue coding molecule, and when we sequence the DNA, we are converting that analogue code into digital code; the 1s and 0s in the computer are very similar to the dots and dashes of Schrodinger's metaphor. I call this process "digitizing biology".
Genomics researcher J. CRAIG VENTER is regarded as one of the leading scientists of the 21st century, most notably for the first sequencing and analysis of the human genome published in 2001 and the most recent and most complete sequencing of his diploid human genome in 2007. He is Co-Founder, Chairman, Synthetic Genomics, Inc.; Founder, J. Craig Venter Institute; Author, A Life Decoded.
J. Craig Venter's Edge Bio Page
A NEW KIND OF SOCIO-INSPIRED TECHNOLOGY
Dirk Helbing [6.19.12]

There's a new kind of socio-inspired technology coming up, now. Society has many wonderful self-organization mechanisms that we can learn from, such as trust, reputation, culture. If we can learn how to implement that in our technological system, that is worth a lot of money; billions of dollars, actually. We think this is the next step after bio-inspired technology.
PROFESSOR DIRK HELBING is Chair of Sociology, in particular of Modeling and Simulation, at ETH Zurich – Swiss Federal Institute of Technology, and the Scientific Coordinator of the FuturICT Flagship Proposal.
TURIN, TUESDAY, JULY 10
THE PERMANENT COLLECTION


Jennifer Jacquet & Ginevra Elkann, President, Pinacoteca Giovanni e Marella Agnelli
EDGE DINNER IN TORINO
Ginevra Elkann e Carlo Antonelli
hanno il piacere di invitarla all'
Edge dinner
in onore di John Brockman, Craig Venter e Brian Eno
martedì 10 luglio
ore 19.30 aperitivo
ore 20.30 cena
Ristorante Del Cambio – Piazza Carignano, 2 – Torino

After Dinner Talk:
J. CRAIG VENTER ANNOUNCES "THE DIGITAL BIOLOGICAL CONVERTER: BIOLOGY AT THE SPEED OF LIGHT"

DINNER HOSTS
Ginevra Elkann, Film Producer, Asmara Films; President, Pinacoteca Giovanni e Marella Agnelli
ATTENDEES
Massimo Banzi, Co-founder, Arduino Project
Gabriele Beccaria, Editor, Tutto Scienza, science supplement of La Stampa
Tommaso Bertani, DJ
Vittorio Bo, Director, Genoa Science Festival; Codice Publishing
John Brockman, Publisher & Editor, Edge.org; CEO, Brockman, Inc.; Author.
Mario Calabresi, Italian Journalist and Author; Director, La Stampa
Andrea Cane, Publishing Director, Trade Division, De Agostini Editore
Max Casacci, Guitarist, Producer
Franca De D'Agostini, Philosopher, University of Turin & University of Milan
Alain Elkann, Novelist, Journalist; President, Egyptian Museum of Turin; Director of Cultural Programs, Italian Television
Brian Eno, Artist; Composer; Recording Producer: U2, Coldplay, Talking Heads, Paul Simon; Recording Artist
Lara Favaretto, Artist
Marco Gilli, Director Politecnico di Torino; Professor, Department of Electronics and Telecommunication
Jennifer Jacquet, Researcher, NYU, studying the effect of honor and shame on cooperation
Heather Kowalski, Communications Consultant, J. Craig Venter Institute
Arto Lindsay, Pop Musician, Audio Provocateur, Producer
Katinka Matson, Artist; Literary Agent; President, Brockman, Inc.; Co-Founder, Edge.org
Marzia Migliora, Artist
Martina Mondadori, Publisher, Tar magazine; Non-Executive Member, Board of Directors, Momdadori
Franco Noero, Galleria Franco Noero
Marcella Pralormo, Director, Pinacoteca Agnelli
Gaetano Prisciantelli, Journalist, Il Venredi, La Rebbublica
Patrizia Sandretto Re Rebaudengo, Director, Fondazione Sandretto Re Rebaudengo
Tadjbakhsh Shahriar, COO, EXOR; Former Director, Goldman Sachs, Paris
Gianluigi Ricuperati, Writer and Essayist, La Repubblica
Scarlett Rouge, Artist
J Craig Venter, Genomics Researcher; Synthetic Genomics, Inc.; J. Craig Venter Institute; Author, A Life Decoded
J. Craig Venter & Ginevra Elkann
DUBLIN, THURSDAY, JULY 12
To celebrate the 70th anniversary of Schroedinger's famous lecture, J. Craig Venter speaks at Examination Hall, Trinity College, Dublin on

[CLICK HERE FOR TEMPORARY PRE-PUBLICATION VIDEO LINK]
[AUDIO]
DUBLIN, SATURDAY, JULY 14
CRAIG VENTER DELIVERS KEYNOTE SPEECH AT EUROPEAN SCIENCE OPEN FORUM, DUBLIN
Ginevra Elkann e Carlo Antonelli
hanno il piacere di invitarla all'
Edge Dinner
in onore di John Brockman, J. Craig Venter e Brian Eno
martedi 10 Luglio
ore 19.30 aperitivo
ore 20.30 cena
Ristorante Del Cambio – Piazza Carignano, 2 – Torino
After Dinner Talk:
J. Craig Venter: "Biology At The Speed Of Light"
DINNER HOSTS
Ginevra Elkann, Film Producer, Asmara Films; President, Pinacoteca Giovanni e Marella Agnelli
Carlo Antonelli, Editor in Chief, Wired Magazine, Italy
ATTENDEES
Massimo Banzi, Co-founder, Arduino Project
Gabriele Beccaria, Editor, Tutto Scienza, science supplement of La Stampa
Tommaso Bertani, DJ
Vittorio Bo, Director, Genoa Science Festival; Codice Publishing
John Brockman, Publisher & Editor, Edge.org; CEO, Brockman, Inc.; Author.
Mario Calabresi, Italian Journalist and Author; Director, La Stampa
Andrea Cane, Publishing Director, Trade Division, De Agostini Editore
Max Casacci, Guitarist, Producer
Franca De D'Agostini, Philosopher, University of Turin & University of Milan
Alain Elkann, Novelist, Journalist; President, Egyptian Museum of Turin; Director of Cultural Programs, Italian Television
Brian Eno, Artist; Composer; Recording Producer: U2, Coldplay, Talking Heads, Paul Simon; Recording Artist
Lara Favaretto, Artist
Marco Gilli, Director Politecnico di Torino; Professor, Department of Electronics and Telecommunication
Jennifer Jacquet, Researcher, NYU, studying the effect of honor and shame on cooperation
Heather Kowalski, Communications Consultant, J. Craig Venter Institute
Arto Lindsay, Pop Musician, Audio Provocateur, Producer
Katinka Matson, Artist; Literary Agent; President, Brockman, Inc.; Co-Founder, Edge.org
Marzia Migliora, Artist
Martina Mondadori, Publisher, Tar magazine; Non-Executive Member, Board of Directors, Momdadori
Franco Noero, Galleria Franco Noero
Marcella Pralormo, Director, Pinacoteca Agnelli
Gaetano Prisciantelli, Journalist, Il Venredi, La Rebbublica
Patrizia Sandretto Re Rebaudengo, Director, Fondazione Sandretto Re Rebaudengo
Tadjbakhsh Shahriar, COO, EXOR; Former Director, Goldman Sachs, Paris
Gianluigi Ricuperati, Writer and Essayist, La Repubblica
Scarlett Rouge, Artist
J Craig Venter, Genomics Researcher; Synthetic Genomics, Inc.; J. Craig Venter Institute; Author, A Life Decoded